BFI INNOVATION
FULL STACK
An end to end platform connecting ideation, prototyping, validation, and market adoption to accelerate clinically validated healthcare innovations
Nationwide research organizations are joining hands to strengthen BFI’s vision of delivering end-to-end support for Indian biomedical innovators

startups
Accelerate & scale 200+ startups nationwide to support healthcare innovation.

Ecosystem
Backed by a $50+ million commitment to accelerate India’s MedTech ecosystem.

Fellowship
Empower 200 fellows with sustained financial support and strategic guidance.
health districts
Bring together infrastructure platforms, 20+ medical colleges in multiple districts.

network
Build a unified commercialization network across major cities.
The composition
bfi innovation
full stack
%20(1).png)
.png)
%20(1).png)
.png)
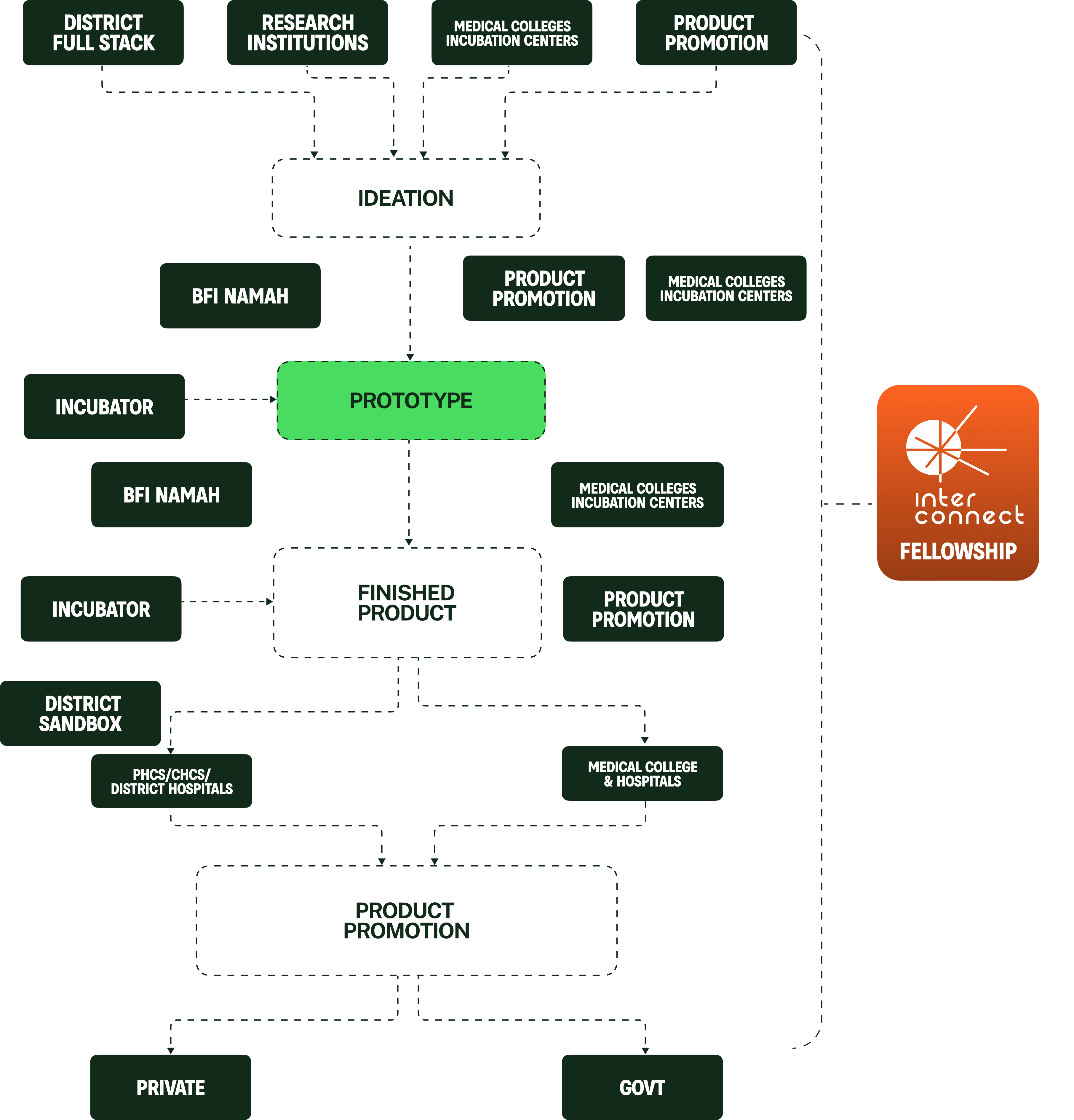
THE Blueprint
How Bfi innovation
full stack works?
.png)
.png)
.png)
.png)
.png)
.png)
.png)
HUB & SPOKE MODEL
The BFI NAMAH will accelerate biomedical innovations from proof-of-concept to market through engineering translation, simulation led design, pilot manufacturing, clinical validation, and regulatory readiness.
.png)
Northen COHORT
Onboarded and Proposed Partners
• ICMR-DHR-mPragati, IIT Delhi
• RCB BioNEST, Faridabad
• AIIMS, New Delhi
• VMMC & Safdarjung Hospital
• GIMS, Greater Noida
• Bareilly International University
• NIMS, Jaipur
• KD University, Mathura
• ICMR-DHR-mPragati, IIT Delhi
• RCB BioNEST, Faridabad
• AIIMS, New Delhi
• VMMC & Safdarjung Hospital
• GIMS, Greater Noida
• Bareilly International University
• NIMS, Jaipur
• KD University, Mathura
western COHORT
Onboarded and Proposed Partners
• Venture Center, Pune
• ACTREC, Mumbai
• AFMC, Pune
• Sassoon Hospital
• DY Patil Hospital, Pune
•Jehangir Hospital, Pune
• Venture Center, Pune
• ACTREC, Mumbai
• AFMC, Pune
• Sassoon Hospital
• DY Patil Hospital, Pune
•Jehangir Hospital, Pune
Southern COHORT
Onboarded and Proposed Partners
• IKP Knowledge Park
• St. John’s Hospital, Bengaluru
• KAHER, Belgaum
• GITAM, Visakhapatnam
• AIG Hospital, Hyderabad, Telangana
• IKP Knowledge Park
• St. John’s Hospital, Bengaluru
• KAHER, Belgaum
• GITAM, Visakhapatnam
• AIG Hospital, Hyderabad, Telangana
.svg)



.png)
.png)
.png)
.png)

